Pharmaceutical Production Facility Engineering
-
Others
From the beginning to the goal of a pharmaceutical facility construction
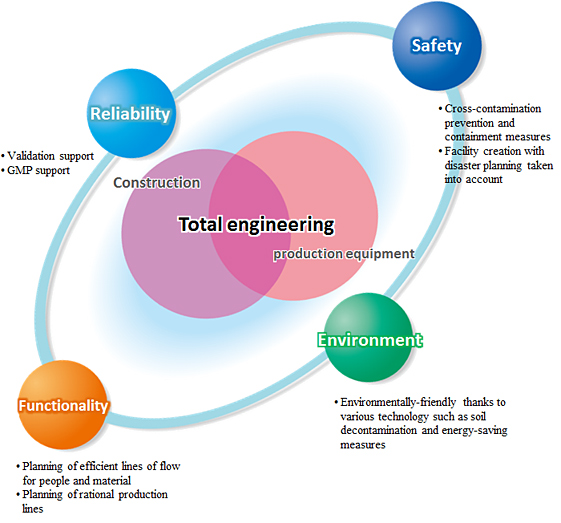
Obayashi Group will provide total support for our client's projects, utilizing our rich know-how and reliable technology in order to achieve stable production as well as high quality and safety that meet international standards.
The merits of comprehensive service
Improved quality
Quality is improved through total quality management whereby all processes from planning, design and construction to maintenance are implemented under a consistent system.
Shortening of the overall process, project cost reduction
Being able to unify the management of various areas of expertise renders coordination between distributors by the client unnecessary and allows shortening of the overall process, resulting in reduced costs and a reduction in the number of client's staff for the project.
Economical cost allocation
Economical cost allocation is possible as the entire process from planning to maintenance is managed via a consistent system.
We will realize the pharmaceutical facility that meets all aspects of the owner's expectations: functionality, durability, safety and sustainability.
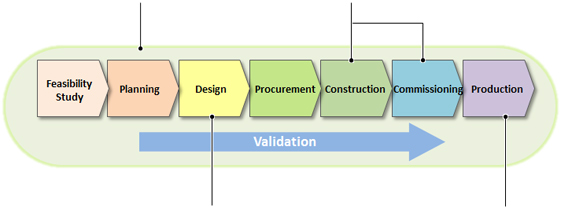
Project development flow
Project management |
Comprehensive supervision on site |
|
Comprehensive coordination during project development. Abundant management technologies will be applied.
|
Various advantages are achieved by comprehensive supervision on site.
|
|
 |
||
Rational design |
After-sales service |
|
Rational and well-balanced design is accomplished taking each design element into account.
|
Long term maintenance of facility is planned by achieving periodic inspection. Extensive organization for maintenance is provided.
|
|
Scope of Obayashi Group Service
We will offer engineering, procurement, construction / installation, commissioning / validation and maintenance services of not only building, electrical and HVAC but also process equipment, purified water system, process utility and material handling system. We realize effective facility by harmonizing building with production equipment
Production equipment

Preparation equipment
We will construct a production system tailored to the client's various needs to facilitate the production of high-quality pharmaceutical products with greater functionality. We boast rich know-how and extensive experience with biological products, injections, solid dosage forms and API(active pharmaceutical ingredients), etc.
Process water/utility equipment

Purified water System
We provide process water that complies with the strict GMP quality control standards. Utility equipment such as clean steam and compressed air, etc. is constructed under a system optimized for the quality that is required.
Automation/In-plant logistics/MES
We implement systems such as production automation systems aimed at the reduction of labor and prevention of erroneous operation, in-plant logistics systems for easy inventory management and streamlined conveyance and manufacturing execution systems aimed at integrating production information management and production support management, etc.

Auto Guided Transport System

Palletizer
Obayashi Group's Technology to Support Each Phase
Plant survey

3-D wind speed measurement
We implement various surveys such as GMP diagnosis, comprehensive plant diagnosis and environmental measurement of the manufacturing area, etc. which are then reflected in the planning for the new plant or plant reconstruction.
Simulation
We will derive optimal solutions utilizing the latest simulation technology, visualize these solutions, making them easy to understand, and incorporate them into the plant design.
BIM (Building Information Modeling) |
Air flow planning by computer simulation |
|
The maintainability of piping and equipment can be examined from different angles and perspectives. |
The most appropriate air flow conditions are determined by using numerical simulation by super computer. The result is then applied to the HVAC design. For example, the simulation such as converting a staircase into a clean room. Another example would be the ventilation caused by the heat emissions from equipment. |
|

3D-CAD |
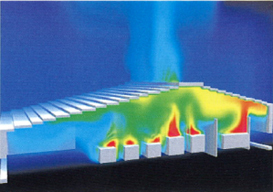
Air Flow Analysis |
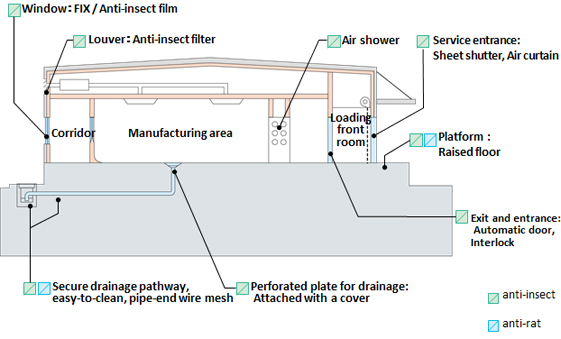
Anti-insect/rodent measures
We provide comprehensive anti-insect/rodent measures that follow the three general rules:
"do not attract them", "do not let them infest the plant" and "prevent breeding and proliferation".
ISS / ceiling panel system that supports walking

ISS:InterStitial Space
Insulation panels that support walking are adopted as ceiling materials which results in increased operability of interstitial spaces. Our unique HEPA filter box and lighting fixture which can be maintained from above the ceiling without contaminating the manufacturing space.
Containment Facility
Facilities in which hazardous materials are handled are required to have containment system of equipment and facilities through their lifecycle. Obayashi Group will provide engineering services to realize the most appropriate facility matching its character by following approach.
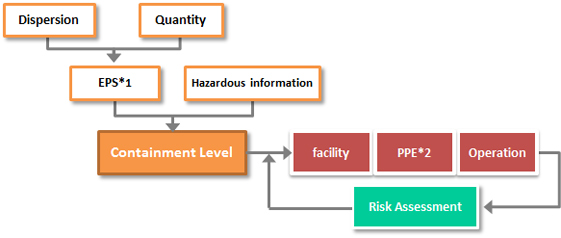
*1 EPS: Exposure Predictor Solid Band
*2 PPE: Personal Protective Equipment
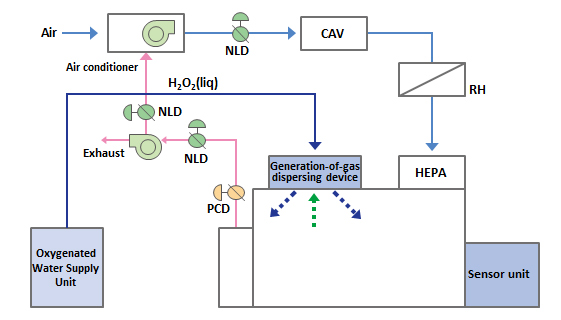
Room Decontamination
Obayashi Group has technologies of room decontamination with Hydrogen Peroxide (H2O2) to decontaminate process rooms and equipment certainly with more attention to prevent corrosion. We will provide the most appropriate system as a facility's character.
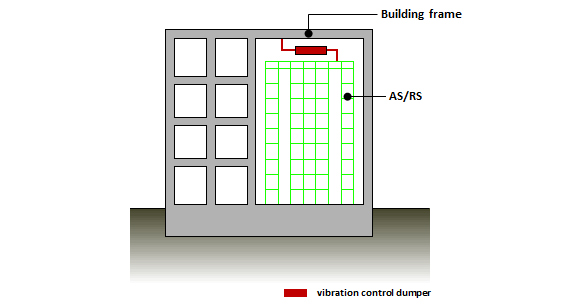
Connecting Type Vibration Control Technology for AS/RS*
*AS/RS: Automated Storage and Retrieval System
This technology controls vibration of building frame and storage rack which are connected with vibration control damper. It prevents dropping stocks and also controls the earthquake input to the building frame.
Other technology we possess
- Containment of hazardous substances
- Cross-contamination prevention measures
- Floor concrete cracking control
- Optimization of floor finishing materials
- Waste water treatment system construction
- Soil contamination countermeasures
- Energy-saving measures
- CO2 emission-reducing technology
- Natural energy-utilizing technology
- Fire and explosion countermeasures
- Disaster countermeasures
(Seismic isolation, damping, BCP support) - Security measures (facilities, information, personnel)
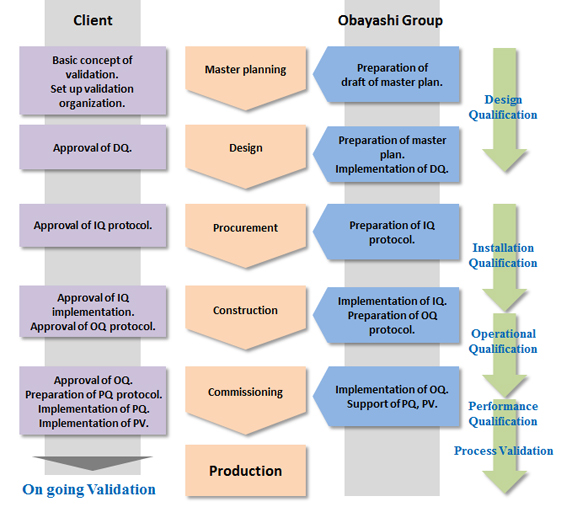
Validation Support
Obayashi Group has a lot of experience to support validation work with fully understanding of GMP. We will totally support owner to develop Validation Master Plan, to develop and implement the DQ/IQ/OQ protocol and to assist the PQ.
Work flow
Major Project

Nichiban Medical Plant No.2
Fukuoka, Japan

Qualicaps Plant No.6
Nara, Japan

Wako Pure Chemical Industries Aichi Plant
Aichi, Japan

Wako Pure Chemical Industries Osaka Plant
Hyogo, Japan

Takeda Healthcare Products Pharmaceutical products warehouse
Kyoto, Japan

Hogy Medical Tsukuba Plant
Ibaraki, Japan

Shiseido Vietnam Inc.
Dong Nai, Vietnam

TOA Pharmaceuticals Nishihongo Plant
Toyama, Japan

Wako Pure Chemical Industries Mie Plant
Mie, Japan

ASGEN Pharmaceutical Mizunami Plant
Gifu, Japan

WAKO Chemical Miyazaki Plant
Miyazaki, Japan

Mochida Pharmaceutical Plant No.1 Expansion
Tochigi, Japan

Measles Vaccine Production Facilities
Hanoi, Vietnam

Japanese Red Cross Society Tatsumi Building
Tokyo, Japan

Assuran Group Tosu Factory
Saga, Japan

The Research Foundation for Microbial Diseases of Osaka University Kanonji Institute
Kagawa, Japan
Oral Solid Dosage Forms

Takeda Healthcare Products Plant No.7
Kyoto, Japan
Ointments / Injections

Mochida Pharmaceutical Plant No.2
Tochigi, Japan

NITTO PHARMACEUTICAL INDUSTRIES, LTD. Ayabe Factory
Kyoto, Japan

Fujisawa Pharmaceutical Toyama Plant
Toyama, Japan

ONO PHARMACEUTICAL Fujiyama Plant
Sizuoka, Japan

Hokkaido Sugar Sapporo Plant Hokkaido, Japan

IONA International Fukushima Plant
Fukushima, Japan


